Not all peptides labeled pure deliver the safety or results you expect. Many athletes and fitness enthusiasts assume a high purity percentage guarantees effectiveness, but hidden impurities can sabotage your performance gains and pose serious health risks. Understanding what purity really means, how it's measured, and what to look for in lab reports is essential to protect yourself and maximize your supplement investment. This guide breaks down peptide purity in clear terms, helping you navigate testing methods, identify red flags, and choose supplements that truly support your goals without compromise.
Table of Contents
- Key takeaways
- What is peptide purity and why does it matter?
- Common impurities in peptides and potential risks
- How to verify peptide purity: interpreting lab tests and COAs
- Practical tips to choose and use high-purity peptides safely
- Explore trusted peptides and suppliers on Pept
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Purity matters for safety | High purity reduces the risk of adverse effects and improves effectiveness. |
| Combined HPLC and MS | Use testing that combines HPLC and MS to reliably identify impurities. |
| Endotoxin testing | Endotoxin testing below 1 EU/mg is essential for injection safety. |
| Target 98 percent purity | Certificate of Analysis should show 98 percent or higher purity for performance claims. |
What is peptide purity and why does it matter?
Peptide purity represents the proportion of your target peptide compared to all other substances in the product. When you purchase a peptide supplement, you want the active compound that delivers results, not a mixture filled with synthesis leftovers, chemicals, or biological contaminants. High purity means more of what you paid for and less of what could harm you or diminish effectiveness.
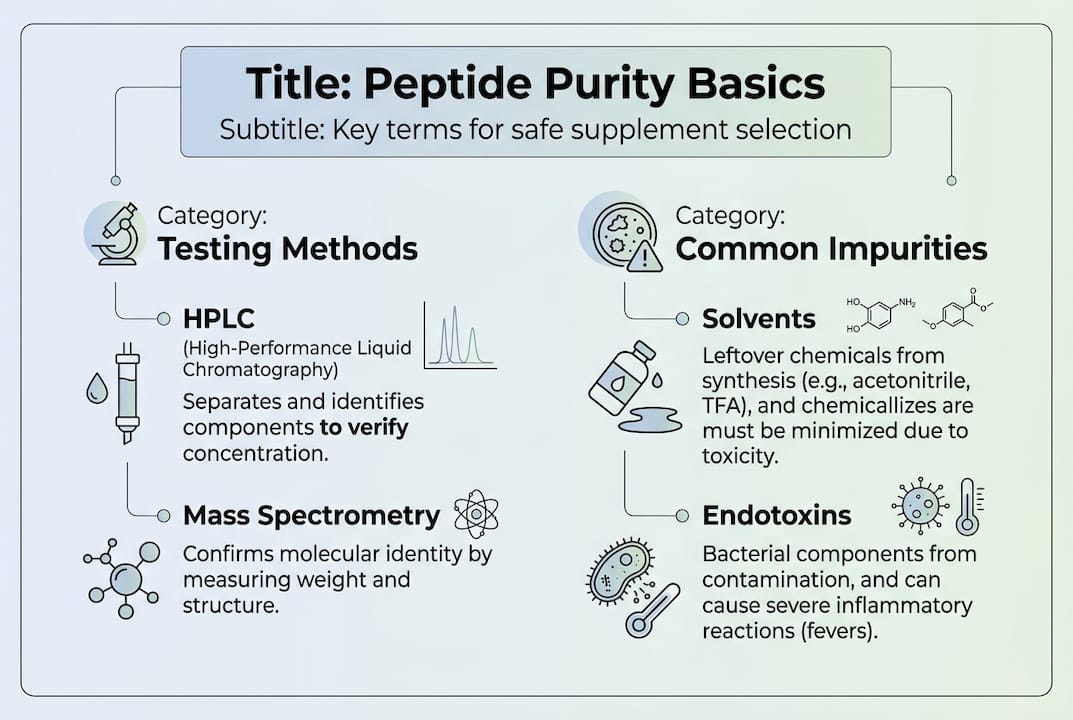
Two primary methods assess peptide purity: High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS). HPLC separates compounds based on their chemical properties and measures the target peptide's concentration relative to other substances. The result appears as a chromatogram showing peaks, where a single dominant peak indicates high purity. MS takes analysis further by confirming the molecular weight matches your target peptide exactly, catching impurities HPLC might miss.
Relying on HPLC alone creates blind spots. This method only detects compounds that absorb UV light, missing non-UV absorbing chemicals and biological contaminants entirely. For athletes injecting peptides, this gap poses serious risks. Endotoxins from bacterial contamination don't show up on standard HPLC tests but can trigger fever, inflammation, and immune system disruption. A peptide purity testing methods shows athletes should prioritize products with combined HPLC, MS, and endotoxin testing at 98% or higher purity.
Why does this matter for your performance and health? Impurities can cause unexpected side effects ranging from injection site reactions to systemic inflammation that undermines recovery. Lower purity peptides may contain inactive or partially formed sequences that compete with the active compound, reducing effectiveness. When you're investing time, money, and effort into optimizing performance, using verified high-purity peptides ensures you get the results you're working toward.
Understanding basic peptide terminology guide helps you communicate effectively with suppliers and interpret technical documentation. The more informed you are, the better equipped you become to spot quality products and avoid substandard options.
Pro Tip: Always review the Certificate of Analysis for chromatograms showing a single dominant peak above 95%, ideally 98% or higher. Multiple peaks or broad, irregular patterns indicate significant impurities that compromise both safety and effectiveness.
Key purity considerations for athletes:
- Verify combined HPLC and MS testing, not HPLC alone
- Check endotoxin levels stay below 1 EU/mg for injection safety
- Prioritize 98% or higher purity for performance applications
- Request independent third-party lab verification
- Examine chromatograms for clean, single-peak patterns
Common impurities in peptides and potential risks
Even peptides marketed as high purity can harbor contaminants that testing methods fail to catch. Synthetic by-products from manufacturing represent the most common impurities, including truncated sequences, deletion peptides, and chemical reagents used during synthesis. These leftovers may seem harmless but can trigger immune responses or interfere with the target peptide's mechanism of action.
Non-UV absorbing chemicals present a hidden danger because standard HPLC testing misses them completely. Solvents, salts, and certain organic compounds used in synthesis don't show up on chromatograms, yet they remain in the final product. For athletes using peptides regularly, accumulated exposure to these invisible contaminants can cause unpredictable reactions or long-term health concerns.
Biological contaminants pose the most immediate risk for injection users. Endotoxins, which are fragments of bacterial cell walls, can contaminate peptides during production or handling. Even trace amounts trigger powerful immune responses, causing fever, fatigue, inflammation, and flu-like symptoms that derail training and recovery. The third-party testing importance confirms HPLC misses these dangerous compounds, and endotoxin levels exceeding 1 EU/mg create significant injection risks.

Heavy metals like lead, mercury, and cadmium can leach into peptides from manufacturing equipment or raw materials. While present in small quantities, chronic exposure through regular supplementation accumulates in tissues over time, potentially affecting neurological function, kidney health, and overall performance. Athletes pushing their bodies hard cannot afford these hidden burdens.
How do impurities affect athletic performance? Beyond direct health risks, contaminants reduce the effective dose of active peptide you receive. A product labeled as 10mg at 90% purity only delivers 9mg of active compound, with 1mg of unknown substances. Over time, this dosing inconsistency makes it impossible to dial in optimal protocols or track results accurately. Impurities can also cause localized inflammation at injection sites, creating discomfort and potentially limiting training intensity.
"Third-party testing is non-negotiable for serious athletes. Supplier-provided COAs may look impressive, but independent verification catches the contaminants that standard methods miss. The difference between verified purity and assumed purity can mean the difference between progress and setbacks."
Understanding peptide safety regulations helps you recognize which standards apply to research peptides versus pharmaceutical-grade products. This knowledge empowers you to ask better questions and demand appropriate documentation from suppliers.
Common peptide contaminants and their sources:
- Truncated sequences from incomplete synthesis
- Chemical reagents and solvents used in production
- Endotoxins from bacterial contamination
- Heavy metals from equipment or raw materials
- Salt residues and buffer compounds
- Degradation products from improper storage
How to verify peptide purity: interpreting lab tests and COAs
Reading a Certificate of Analysis transforms from intimidating to empowering once you understand what each section reveals. The HPLC chromatogram appears as a graph with peaks representing different compounds detected in the sample. Your target peptide should produce a single, sharp, dominant peak, typically labeled with the retention time and purity percentage. Multiple peaks, especially those close in size to the main peak, indicate significant impurities that reduce product quality.
The Mass Spectrometry spectrum confirms molecular identity by showing the exact molecular weight of compounds present. This section should display a clear match between the observed mass and the theoretical mass of your target peptide. Even a small deviation suggests the wrong compound, a modified sequence, or contamination. MS data catches problems HPLC misses, making it an essential verification layer.
Endotoxin reports measure bacterial contamination using the Limulus Amebocyte Lysate (LAL) test, expressed in Endotoxin Units per milligram (EU/mg). For injection-grade peptides, levels must stay well below 1 EU/mg. Higher readings indicate bacterial contamination during manufacturing or handling, posing serious infection and inflammation risks. This single number can make the difference between safe use and dangerous complications.
Pro Tip: Prioritize peptides verified by independent third-party labs rather than supplier-only COAs. Independent testing eliminates conflicts of interest and provides unbiased results you can trust when making health decisions.
Step-by-step COA verification process:
- Confirm the COA matches the product batch number exactly
- Check the testing date is recent, ideally within 6 months
- Verify the lab performing tests is independent and accredited
- Examine the HPLC chromatogram for a single dominant peak
- Confirm purity percentage meets or exceeds 98%
- Review MS data shows molecular weight matches target peptide
- Check endotoxin levels remain below 1 EU/mg
- Look for heavy metal testing results if available
- Verify the document includes lab contact information
- Cross-reference supplier claims with actual test results
| Parameter | Acceptable for Athletes | Non-Acceptable | Risk Level |
|---|---|---|---|
| HPLC Purity | ≥98% | <95% | High if below 95% |
| MS Match | Exact molecular weight | >1 Da deviation | Critical |
| Endotoxin Level | <0.5 EU/mg | >1 EU/mg | Severe for injection |
| Chromatogram | Single sharp peak | Multiple peaks | Moderate to high |
| Testing Date | <6 months old | >12 months old | Moderate |
| Lab Type | Independent third-party | Supplier-only | Moderate |
Authenticity verification matters because falsified COAs circulate in the peptide market. Contact the testing lab directly using information from their official website, not details provided by the supplier. Ask them to confirm they tested the specific batch and issued the COA. Legitimate labs maintain records and can verify documents quickly. If a supplier resists this verification or the lab has no record, walk away immediately.
Understanding proper peptide storage guide becomes crucial once you verify purity, because even high-quality peptides degrade without correct handling. The interpreting peptide COAs resource confirms checking for clean chromatograms, MS matches, and low endotoxin levels while avoiding anything under 95% purity for performance applications.
Practical tips to choose and use high-purity peptides safely
Sourcing decisions start with supplier credibility. Research companies thoroughly by checking how long they've operated, reading independent reviews from verified users, and confirming they provide detailed COAs for every batch. Reputable suppliers welcome questions about testing methods and readily share documentation. Companies that dodge inquiries about purity testing or provide vague answers likely cut corners that compromise your safety.
Verify testing certifications by confirming suppliers use accredited independent labs, not just in-house testing. Look for ISO/IEC 17025 accreditation, which ensures labs meet international standards for technical competence. Request batch-specific COAs rather than generic certificates that may not represent the actual product you receive. Each batch can vary in purity, making individual verification essential.
Storage conditions directly impact peptide purity over time. Lyophilized (freeze-dried) peptides remain stable at -20°C for extended periods, but exposure to heat, light, or moisture triggers degradation. Once reconstituted, peptides become much more fragile and typically require refrigeration at 2-8°C with use within 30 days. Improper storage transforms a pure peptide into a degraded mixture that loses effectiveness and potentially forms harmful breakdown products.
Safe handling prevents contamination that lab testing cannot predict. Always use sterile technique when reconstituting and drawing peptides, including alcohol swabs, sterile vials, and proper needle handling. Never reuse needles or share vials between individuals. Contamination introduces bacteria that produce endotoxins, creating the exact risks you avoided by choosing pure peptides initially.
Pro Tip: Always choose peptides tested by independent labs with clear COA transparency. If a supplier cannot provide batch-specific third-party testing results, their purity claims are unverified and potentially misleading.
Reliable suppliers distinguish themselves through consistent quality, transparent testing, and responsive customer service. The vetted peptide suppliers directory connects you with companies meeting strict purity standards and providing verified documentation. Avoid products with unclear purity claims, missing COAs, or prices significantly below market rates, which often indicate compromised quality.
Practical checklist for safe peptide use:
- Research supplier reputation through independent reviews and community feedback
- Verify independent third-party testing for every batch purchased
- Confirm purity meets or exceeds 98% with combined HPLC, MS, and endotoxin testing
- Store lyophilized peptides at -20°C in original sealed vials
- Refrigerate reconstituted peptides at 2-8°C and use within 30 days
- Use sterile technique for all reconstitution and injection procedures
- Never share vials or reuse needles between doses
- Track batch numbers and correlate with results for quality consistency
- Monitor for unexpected reactions that may indicate contamination
- Dispose of expired or improperly stored peptides safely
The peptide purity recommendations emphasize athletes should prioritize products with 98% or higher purity verified through HPLC, MS, and endotoxin testing from trusted third-party sources. This standard protects your health while ensuring you receive the performance benefits you're investing in.
Explore trusted peptides and suppliers on Pept
Now that you understand peptide purity and how to verify quality, finding reliable sources becomes your next step. Pept serves as a comprehensive platform connecting athletes and fitness enthusiasts with vetted peptide suppliers who meet strict purity and testing standards. Rather than navigating the market blindly, you can explore pre-screened options that prioritize your safety and results.
Browse the vetted peptide suppliers directory to discover companies providing transparent third-party testing, detailed COAs, and consistent quality. Suppliers like Royal Peptides supplier exemplify the standards you should expect, offering verified purity and responsive support for your questions. The research peptide directory helps you explore specific compounds with clear information about their applications, dosing considerations, and quality requirements, making informed decisions straightforward and confidence-building.
Frequently asked questions
What is peptide purity and how is it measured?
Peptide purity represents the percentage of target peptide versus all other substances in a product. It's measured primarily through High-Performance Liquid Chromatography (HPLC), which separates compounds and quantifies the target peptide, and Mass Spectrometry (MS), which confirms molecular identity. Combined testing provides the most accurate purity assessment.
Why is 98% purity recommended for athletes?
Purity at 98% or higher minimizes exposure to impurities that can cause side effects, reduce effectiveness, or pose health risks. Lower purity means more contaminants per dose, increasing the chance of adverse reactions and inconsistent results. For injection use, higher purity standards become critical for safety.
Can HPLC testing alone guarantee peptide safety?
No, HPLC only detects UV-absorbing compounds and misses biological contaminants like endotoxins and non-UV absorbing chemicals. Comprehensive safety requires combined HPLC, Mass Spectrometry, and endotoxin testing. Relying on HPLC alone leaves dangerous blind spots in quality verification.
How do I verify a Certificate of Analysis is legitimate?
Contact the testing lab directly using information from their official website to confirm they tested the specific batch and issued the COA. Check that batch numbers match your product, testing dates are recent, and the lab is independent and accredited. Legitimate suppliers welcome verification requests.
What endotoxin level is safe for injectable peptides?
Endotoxin levels should remain below 1 EU/mg for injectable peptides, with levels below 0.5 EU/mg preferred for regular use. Higher levels can trigger immune responses, inflammation, and flu-like symptoms. Always verify endotoxin testing appears on the COA before using peptides via injection.
How does improper storage affect peptide purity?
Improper storage causes peptide degradation, forming breakdown products that reduce purity and effectiveness over time. Heat, light, and moisture accelerate this process. Even high-purity peptides become contaminated mixtures when stored incorrectly, making proper handling essential for maintaining quality.
